Teplizumab
Description
This is a humanized IgG1 monoclonal antibody using the same sequences as the therapeutic antibody teplizumab. It specifically binds to CD3 molecules expressed on the surface of both CD4+ and CD8+ T cells, which are involved in the destruction of pancreatic beta cells. One hypothesis suggests that teplizumab acts as a partial agonist at the T cell receptor (TCR), leading to an increased population of exhausted T cells positive for KLRG1, TIGIT and CD8. These exhausted T cells persist but exhibit impaired effector functions, thereby unlikely to contribute to further β cell destruction. The design incorporates an Fc-non-binding feature aiming to minimize the incidence of cytokine release syndrome (CRS). Teplizumab is indicated to delay the onset of Stage3 type 1 diabetes (T1D) in adults and pediatric patients aged 8 years and older with Stage 2 T1D.
Product name | Teplizumab Biosimilar |
Species | Homo sapiens |
Expression system | CHO-K1 |
Buffer | PBS, pH 7.4 |
Delivery condition | Dry ice (-80°C) |
Delivery Time | 1 week if in stock; 4 weeks if production needed |
Storage condition | Store at -80°C |
Brand | BioMetas |
Applications | ELISA, assay, in vivo |
Aliases/Synonyms | Tzield™, hOKT3-gamma-1 (Ala-Ala), humanized OKT3, MGA031, teplizumab-mzwv |
Reference | |
Note | For research use only. Not suitable for clinical or therapeutic use. |
Isotype | IgG1 |
Clonality | Monoclonal Antibody |
Size | 1mg, 5mg, 10mg, 50mg, 100mg |
Brand | BioMetas |
Product type | Biosimilar |
Clonality | Monoclonal Antibody |
Expression system | CHO-K1 |
Applications | Elisa, assay, in vivo |
| Amount | Price |
| 1mg | ¥1200 |
| 5mg | ¥3000 |
| 10mg | ¥5000 |
| 25mg | ¥7500 |
| 50mg | ¥10000 |
| 100mg | ¥14000 |
Contact Us for a Quote!
Data Gallery
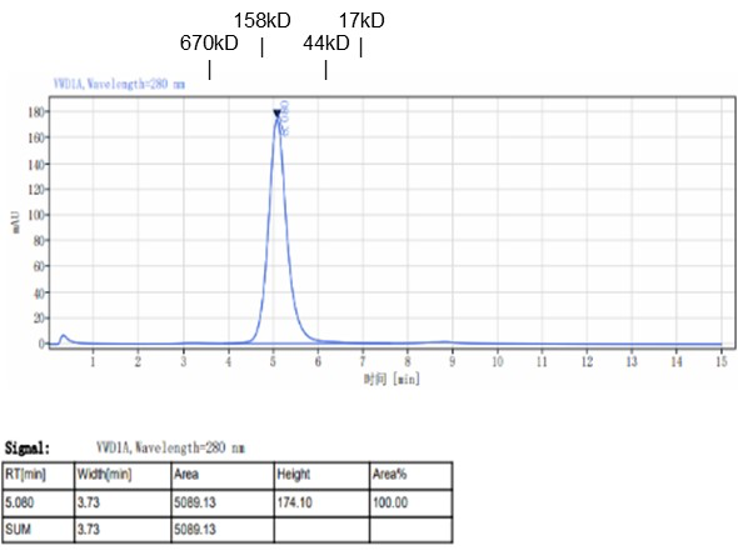
Fig. 1.) 4-20% SDS-PAGE analysis
Recombinant protein was visualized by Coomassie Brilliant Blue R250 staining.
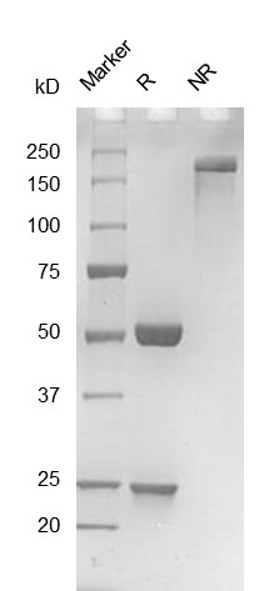
Fig. 2.) SEC-HPLC analysis
Column: Superdex 200 Increase 5/150 GL
Running buffer: 2xPBS, pH 7.4